
The Melting point is 235 degrees Celsius and the boiling point is 178 degrees Celsius. Caffeine C8H10N4O - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. These strong intermolecular forces make it difficult to break the molecule apart, therefore more energy is needed causing a high melting and boiling point. The London force is the immediate attraction of electrons from one atom to the positive nuclei of other surrounding atoms. 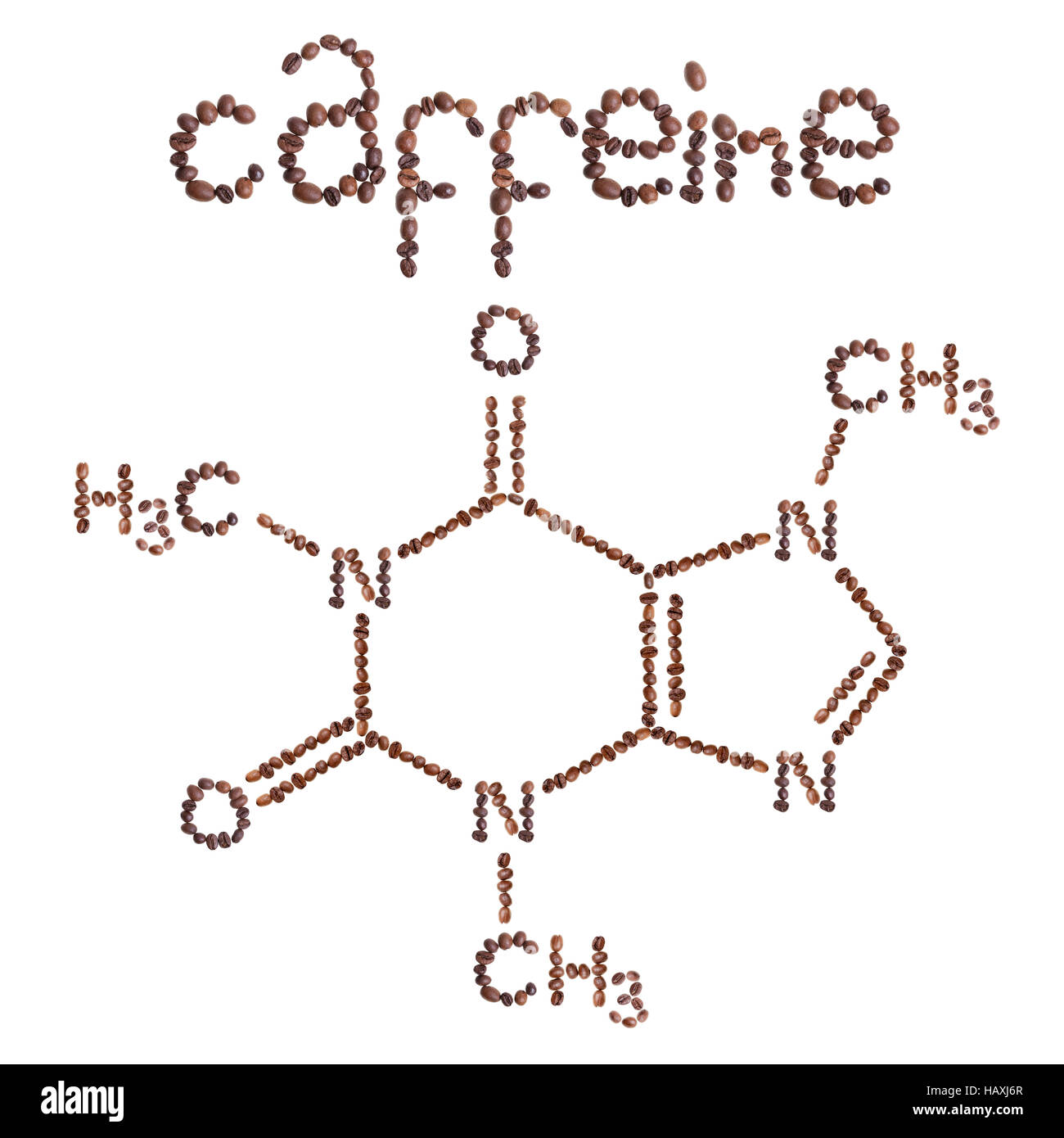
London Dispersion forces exist because the electrons are always in motion, temporarily becoming more positive or negative. Articles of Caffeine are included as well. These charged ends are attracted to other opposite charged ends creating a very strong intermolecular force. Chemsrc provides Caffeine (CAS:58-08-2) MSDS, density, melting point, boiling point, structure, formula, molecular weight etc. These bond dipoles within the molecule create an overall molecular dipole therefore the molecule has one negative end and one positive end. Synonyms : 1,3,7-Trimethylxanthine Coffeinum Melting point/ Freezing point(C), 234 - 236 C pH value, 6.9 Density (g/cm3), 1.23 Partition coefficient Log. They occur whenever there is a separation of positive and negative charges. Caffeine molecules have bond dipoles that measure the polarity of a chemical bond within a molecule. For eachcompound give:Product Name, Chemical Formula, Formula Weight, Melting Point Boiling Point and Density,Health Hazard Data (summarize in your own. The intermolecular forces that are present in caffeine are London dispersion forces and Dipole-dipole forces.ĭipole-Dipole forces exist between the positive end of one polar molecule and the negative end of another polar molecule. Intermolecular forces also known as van der Waals forces consist of Hydrogen bonding, London dispersion forces and Dipole-dipole forces.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
